 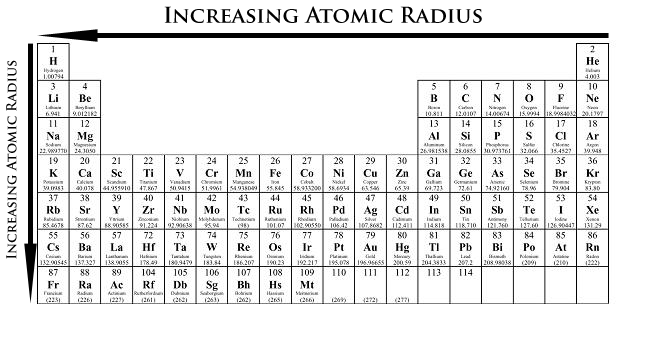
Valence electrons are simultaneously attracted to the positive charge of the nucleus and screened (repelled) by the negative charges of other electrons. Many of the periodic properties of atoms depend on electron configuration in particular, the valence electrons and their level of attraction to the nucleus. :max_bytes(150000):strip_icc()/PeriodicTable_AtomSizes-56a131193df78cf772684720.png)
There may be a few points where an opposite trend is seen, but there is an overall trend when considered across a whole row or down a whole column of the periodic table. There is no other tool in science that allows us to judge relative properties of a class of objects like this, which makes the periodic table a very useful tool. The variation of properties versus position on the periodic table is called periodic trends. 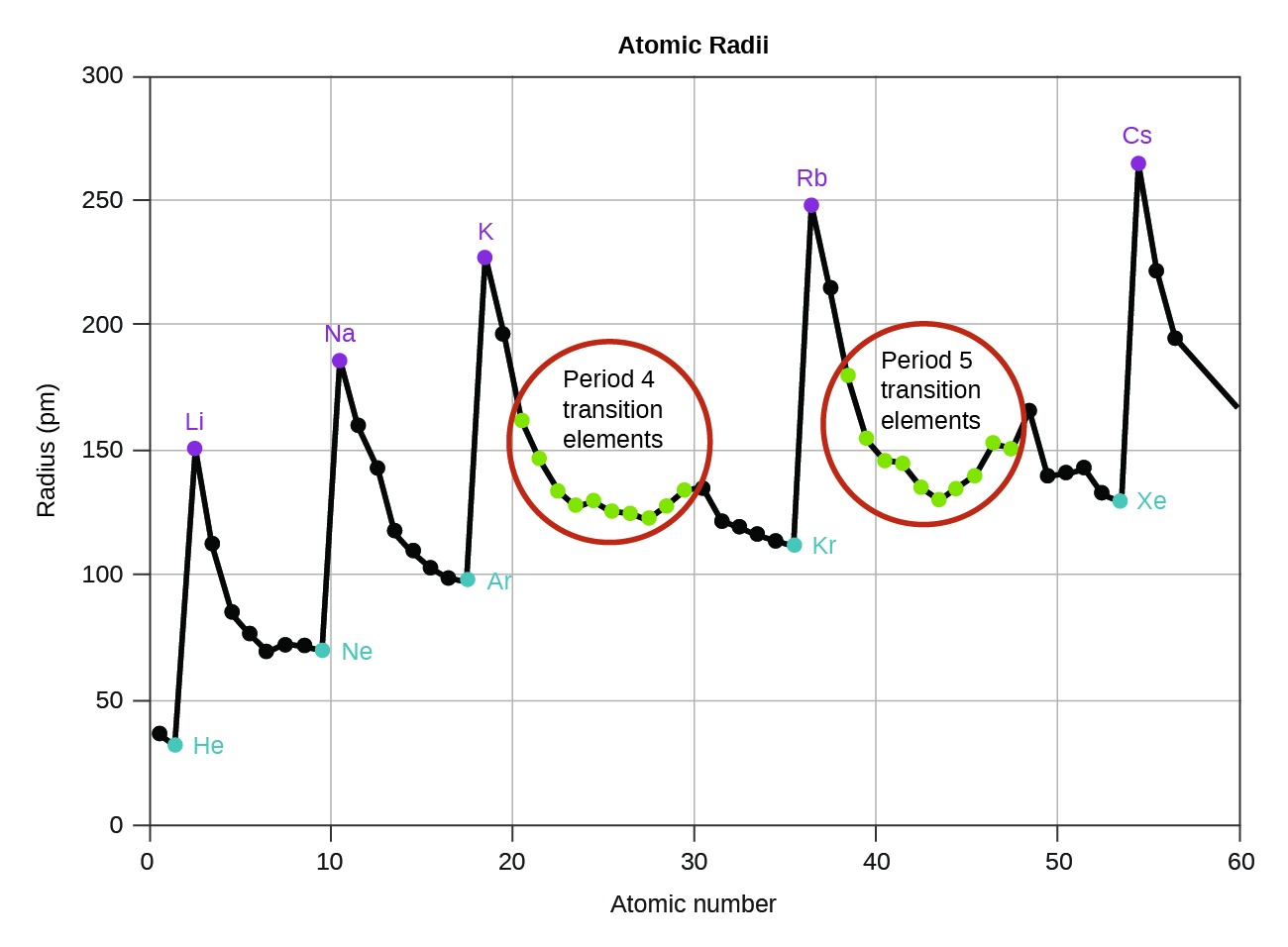
One of the reasons the periodic table is so useful is because its structure allows us to qualitatively determine how some properties of the elements vary versus their position on the periodic table. Be able to state how certain properties of atoms vary based on their relative position on the periodic table.Next, we'll apply the same principles to see if there is any periodic trend for ionic radius. While the energy level would be the predominant factor. In summary, when comparing atomic radii of two elements, consider two factors: So if we draw the atoms on a piece of paper, they may look like something similar to the illustration below. Ultimately, we say atomic radius decreases across the period because of 1) increasing effective nuclear charges, 2) greater attraction between nucleus and valence electrons, and 3) same number of energy levels. Since all elements are having the same number of energy levels, this little movement of valence electrons plays a significant role. The increased attraction would pull the valence electrons closer to the nucleus, and hence a smaller radius. By considering the increasing nuclear charges and unchanged shielding effect, we could easily conclude that the effective nuclear charges increase across the period, and so do the attraction experienced by the valence electrons. Meanwhile, they all have the same number of inner electrons, and hence the same shielding effect. This leads to greater attraction towards the valence electron. Their nuclear charges are increasing from the left to the right of the period. The elements across a period are in the sequence of increasing atomic number. So how does the atomic radius change? Is there any trend in such a case? Let's take a look at the nuclear charge and the shielding effect, and see if we could get an answer. On the other hand, when we travel across the periods from the left to the right, we'll get atoms with the same number of energy levels despite their total numbers of electrons are increasing. So we say, atomic radius increases down the group because of more energy levels. Just imagine yourself wearing more and more clothes, aren't you getting bigger and bigger in size? That's exactly what's happening to the atoms. In practice, we would find out the atomic radius of an atom by measuring the distance between two nuclei of the same element in a molecule and defining half of the distance as the bonding atomic radius (\( r_b = \frac4p^65s^1\) Conventionally, we define a spherical surface that encloses the space where the probability of finding electrons inside is 90% as the boundary of the atom, and its radius as the atomic radius. 
All we have is the probability of finding an electron at a particular position. We can't determine exactly where the electrons are or where they will be. They are more like flashing at various locations. Electrons, however, do not travel like the Earth does. So what's atomic radius? Atomic radiusĪtoms are made of a nucleus at the center and one or more electrons moving around the nucleus. The first periodic trend we'll learn is about the atomic radius. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
